Oral TYK2 Treatment Envudeucitinib Shows Strong Phase 3 Psoriasis Results
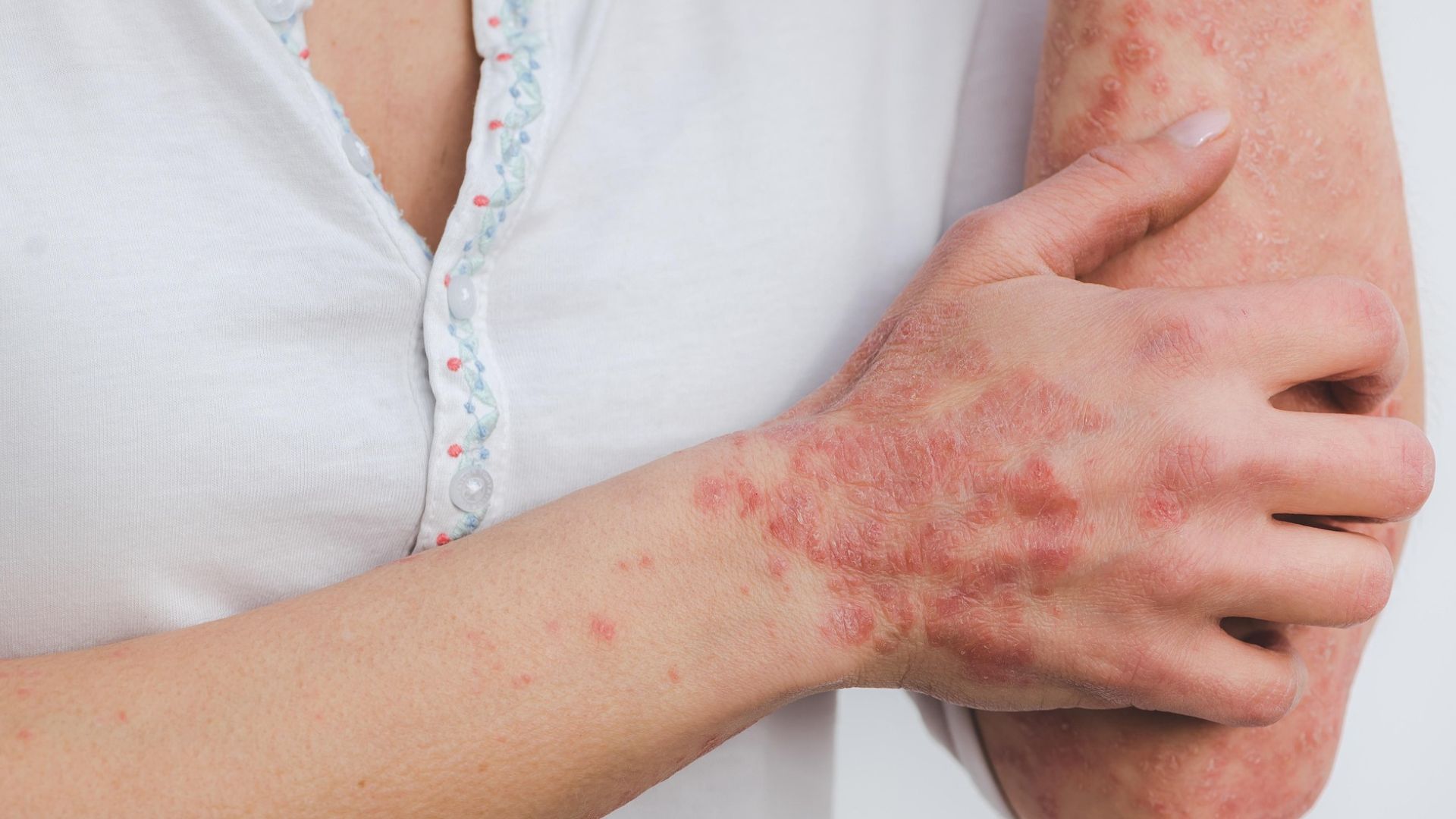
Alumis Inc. has scored a major win in psoriasis drug development with its oral TYK2 inhibitor envudeucitinib, reporting positive outcomes from two Phase 3 clinical trials.
The experimental pill helped patients with moderate-to-severe plaque psoriasis achieve significant skin clearance after four months of treatment, hitting key study goals tied to disease improvement. In these trials, most participants saw at least a 75% reduction in skin lesions, with many judged by physicians to be “clear” or “almost clear.”
What makes these results stand out is that envudeucitinib’s efficacy appears competitive with both emerging oral therapies and established injectable biologics. That could shake up a market dominated by biologics that require injections, offering patients an effective daily pill option.
Following the news, Alumis’ stock more than doubled in value, underscoring investor confidence in the drug’s potential. The company plans to pursue U.S. regulatory approval later this year.
Envudeucitinib joins a growing class of next-generation TYK2 inhibitors being tested for psoriasis and other immune disorders, as drugmakers race to develop safe, effective oral alternatives to current treatments.
If this is something that you and your company are interested in, join Elisa Muscianisi, Vice President, Clinical Development from Alumis Inc.,in her panel discussion on: Modernising Dermatology Trial Design: Reducing Placebo Noise, Updating Endpoints & Strengthening Site Execution.